Publications
Publications categorized by chronological order. Generated by jekyll-scholar.
2025
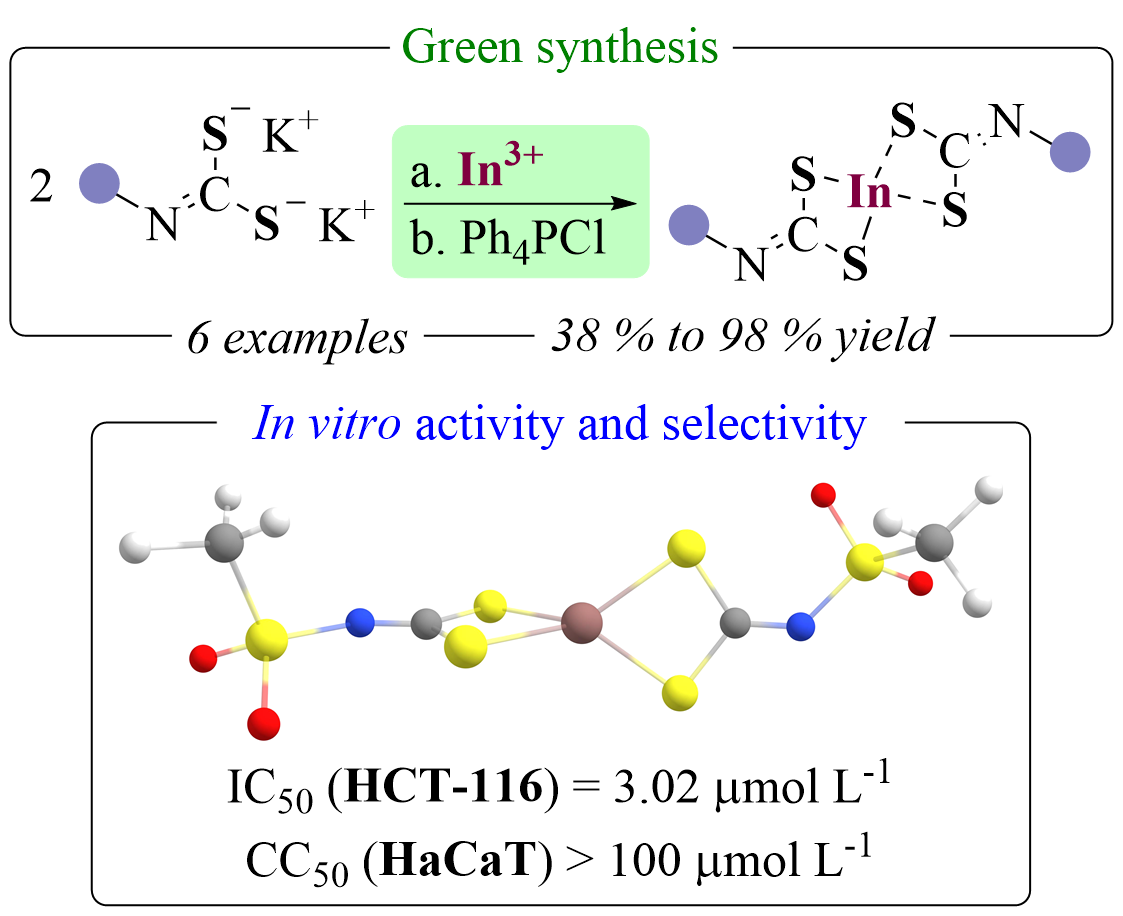
- In(III) complexes of sulfonyldithiocarbimates as selective antineoplasic agents against human colorectal adenocarcinomaLucas Raposo Carvalho*, Aline de Souza Bozzi*, Wanessa Maria Alves Faria, Lucas Gil Venturelli, Túlio Duque Esteves, Sâmia Sousa Duarte, Ramon Ramos Marques Souza, Isione Oliveira Castro, Eclair Venturini Filho, Renata Diniz, Marianna Vieira Sobral, Willian Ricardo Rocha, and Eder do Couto TavaresDalton Transactions, 2025
The synthesis, antineoplasic profile, and structural aspects of six In(III) sulfonyldithiocarbimate complexes (Ph4P[(RSO2N=CS2)2In], 2a–f) are described. The salts were readily obtainable with 38% to 98% yield by the water-mediated complexation of the ligand (1a–f) in the presence of In(NO3)3 without further purification. Spectroscopic data pointed to the formation of isomers, which were postulated as the result of three complexation modes – SS–SS, SN–SN, and SN–SS. DFT calculations furnished ΔG, Keq, and δ values that indicated a preference for the SS–SS isomer. Statistical analysis of 13C NMR data placed 13C=N δ values as efficient probes of the \textitd-electron count of the metal in sulfonyldithiocarbimate complexes. The compounds displayed antineoplasic activity against human colorectal adenocarcinoma (HCT-116) cell lines (IC50 = 3.02(21) μmol L−1 to 5.36(42) μmol L−1) with high selectivity compared to HaCaT cells. Moreover, an XRD analysis of a water-insoluble decomposition product of a ligand (1b) is also described, showing the formation of a supramolecular-like network and corroborating the use of the metallic complexes as biologically active compounds instead of their isolated ligands.
@article{D5DT00868A, author = {Carvalho, Lucas Raposo and Bozzi, Aline de Souza and Faria, Wanessa Maria Alves and Venturelli, Lucas Gil and Esteves, Túlio Duque and Duarte, Sâmia Sousa and de Souza, Ramon Ramos Marques and Castro, Isione Oliveira and Filho, Eclair Venturini and Diniz, Renata and Sobral, Marianna Vieira and Rocha, Willian Ricardo and Tavares, Eder do Couto}, title = {In(III) complexes of sulfonyldithiocarbimates as selective antineoplasic agents against human colorectal adenocarcinoma}, journal = {Dalton Transactions}, year = {2025}, volume = {54}, issue = {29}, pages = {11207-11224}, publisher = {The Royal Society of Chemistry}, doi = {10.1039/D5DT00868A}, dimensions = {true}, }
2023
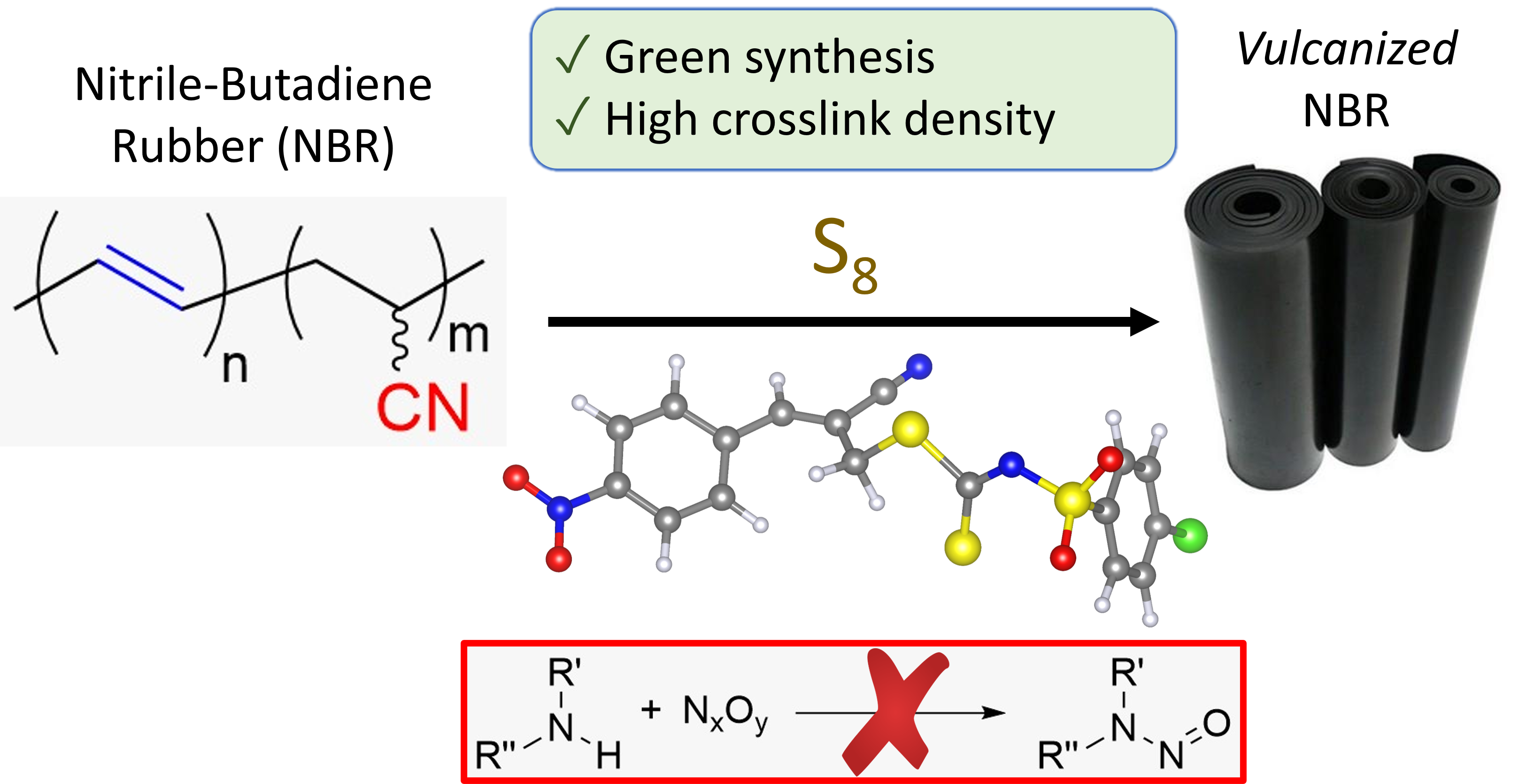
- Tetraphenylphosphonium Allyldithiocarbimates: A Novel Safe Amine Class of Morita–Baylis–Hillman-Derived NBR Vulcanization AcceleratorsLucas Raposo Carvalho*, Lucas Gil Venturelli, Thiago Castro Lopes, Eclair Venturini Filho, Vinicius Gonçalves Souto Fontenelle, Guilherme Ferreira de Lima, Leila Léa Yuan Visconte, and Eder do Couto TavaresACS Sustainable Chemistry & Engineering, 2023
Eliminating \textitN-nitrosamines from rubber products, which are introduced by common vulcanization accelerators, is a significant challenge faced by the industry. The class of tetraphenylphosphonium allyldithiocarbimate salts offers a green and operationally simple alternative to traditional accelerators. This study describes the synthesis of four novel compounds and their application as accelerators for nitrile-butadiene rubber (NBR) vulcanization. The compounds were synthesized from easily accessible Morita–Baylis–Hillman acetates and potassium dithiocarbimate salts using acetone:water (1:1, v:v) as the solvent, which produced a precipitate that was washable with water. Compared to vulcanizates with tetramethyl thiuram disulfide (TMTD), those with the allyldithiocarbimate compounds showed longer t90 times, faster ts1 times, comparable tensile strength, and better tear resistance, indicating that these compounds can be a viable alternative for producing good-quality rubber artifacts.
@article{doi:10.1021/acssuschemeng.3c03586, author = {Carvalho, Lucas Raposo and Venturelli, Lucas Gil and Lopes, Thiago Castro and Filho, Eclair Venturini and Fontenelle, Vinicius Gonçalves Souto and {de Lima}, Guilherme Ferreira and Visconte, Leila Léa Yuan and Tavares, Eder do Couto}, title = {Tetraphenylphosphonium Allyldithiocarbimates: A Novel Safe Amine Class of Morita–Baylis–Hillman-Derived NBR Vulcanization Accelerators}, journal = {ACS Sustainable Chemistry \& Engineering}, volume = {11}, number = {39}, pages = {14507-14517}, year = {2023}, doi = {10.1021/acssuschemeng.3c03586}, dimensions = {true}, } - PatentPeptídeo sintético derivado de filoseptina e usoLucas Raposo Carvalho, Jarbas Magalhães Resende, Thayane Santos de Carvalho, Jaqueline Maria Siqueira Ferreira, and Gabriela Francine Martins LopesOct 2023Patent Application no. BR102023020443A2, filed October 2023. Deposited by UFMG and UFSJ
A presente tecnologia trata de um peptídeo sintético de 22 aminoácidos, aqui identificado como R1G2-PS-O1 (SEQ ID Nº1), derivado do peptídeo filoseptina (UniProtKB: P84569.1). A presente tecnologia também trata do uso de R1G2-PS-O1 na preparação de medicamentos antifúngicos para o tratamento de infecções fúngicas, incluindo candidíases. O peptídeo da presente tecnologia apresenta atividade antifúngica a partir da concentração inibitória mínima (CIM) de 3,35 µM contra cepas de Candida sp (linhagens ATCC e isolados clínicos), além de apresentar efeito sinérgico com o miconazol.
@misc{carvalho2023peptideo, author = {Carvalho, Lucas Raposo and Resende, Jarbas Magalhães and {de Carvalho}, Thayane Santos and Ferreira, Jaqueline Maria Siqueira and Lopes, Gabriela Francine Martins}, title = {Peptídeo sintético derivado de filoseptina e uso}, year = {2023}, month = oct, note = {Patent Application no. BR102023020443A2, filed October 2023. Deposited by UFMG and UFSJ}, }
2022
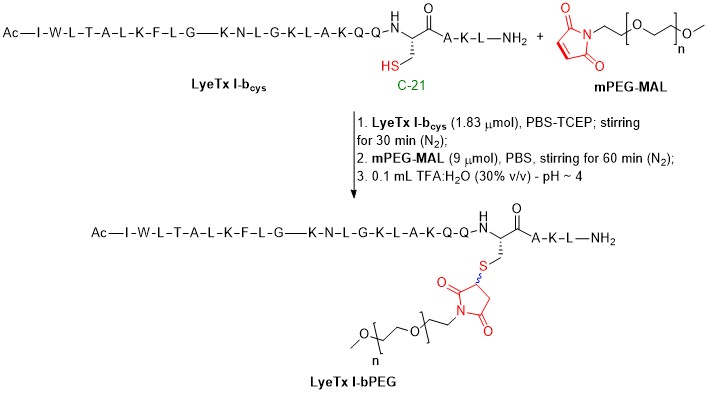
- PEGylation of the antimicrobial peptide LyeTx I-b maintains structure-related biological properties and improves selectivityJúlio César Moreira Brito†, Lucas Raposo Carvalho†*, Amanda Neves de Souza, Guilherme Carneiro, Paula Prazeres Magalhães, Luiz Macêdo Farias, Natália Rocha Guimarães, Rodrigo Moreira Verly, Jarbas Magalhães Resende, and Maria Elena de Lima*Frontiers in Molecular Biosciences, Oct 2022
The biological activity of antimicrobial peptides and proteins is related to their structural aspects and is sensible to post-translational modifications like PEGylation. However, PEGylation of protein and peptide drugs has expanded in recent years due to the reduction of their toxicity. Due to their size, PEGylation can preserve or compromise the overall structure of these biopolymers and, thus biological properties. LyeTx I-bcys was synthesized by Fmoc strategy and coupled to polyethylene glycol 2.0 kDa. The conjugates were purified by HPLC and characterized by MALDI-ToF-MS analysis. Microbiological assays with LyeTx I-bcys and LyeTx I-bPEG were performed against Staphylococcus aureus (ATCC 33591) and Escherichia coli (ATCC 25922) in liquid medium. MIC values of 2.0 and 1.0 µM for LyeTx I-bcys and 8.0 and 4.0 µM for LyeTx I-bPEG were observed against S. aureus and E. coli, respectively. PEGylation of LyeTx I-bcys (LyeTx I-bPEG) decreased the cytotoxicity determined by MTT method for VERO cells compared to the non-PEGylated peptide. In addition, structural and biophysical studies were performed to evaluate the effects of PEGylation on the nature of peptide-membrane interactions. Surface Plasmon Resonance experiments showed that LyeTx I-b binds to anionic membranes with an association constant twice higher than the PEGylated form. The three-dimensional NMR structures of LyeTx I-bcys and LyeTx I-bPEG were determined and compared with the LyeTx I-b structure, and the hydrodynamic diameter and zeta potential of POPC:POPG vesicles were similar upon the addition of both peptides. The mPEG-MAL conjugation of LyeTx I-bcys gave epimers, and it, together with LyeTx I-bPEG, showed clear α-helical profiles. While LyeTx I-bcys showed no significant change in amphipathicity compared to LyeTx I-b, LyeTx I-bPEG was found to have a slightly less clear separation between hydrophilic and hydrophobic faces. However, the similar conformational freedom of LyeTx I-b and LyeTx I-bPEG suggests that PEGylation does not cause significant structural changes. Overall, our structural and biophysical studies indicate that the PEGylation does not alter the mode of peptide interaction and maintains antimicrobial activity while minimizing tissue toxicity, which confirmed previous results obtained in vivo. Interestingly, significantly improved proteolytic resistance to trypsin and proteinase K was observed after PEGylation.
@article{10.3389/fmolb.2022.1001508, author = {Moreira Brito, Júlio César and Carvalho, Lucas Raposo and {Neves de Souza}, Amanda and Carneiro, Guilherme and Magalhães, Paula Prazeres and Farias, Luiz Macêdo and Guimarães, Natália Rocha and Verly, Rodrigo Moreira and Resende, Jarbas Magalhães and {de Lima}, Maria Elena}, title = {PEGylation of the antimicrobial peptide LyeTx I-b maintains structure-related biological properties and improves selectivity}, journal = {Frontiers in Molecular Biosciences}, volume = {9}, year = {2022}, doi = {10.3389/fmolb.2022.1001508}, issn = {2296-889X}, dimensions = {true}, }